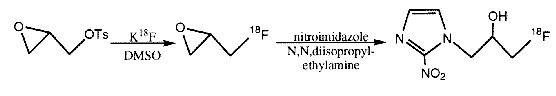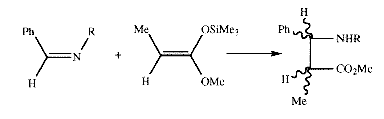|
|
 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Application of Microwaves to Organic Chemistry
© G. Whittaker, 1994 & 2007. This work, or extracts from this work may be reproduced only with the written permission of the author.
ContentsOrganic Syntheses at Atmospheric Pressure Organic Syntheses at Elevated Pressure Is there a microwave specific effect? Organic Syntheses in Dry Media
The popularity of microwave heating for organic synthesis has increased to the extent that it now forms the basis of a number of commercial systems, and has even made its way into undergraduate laboratory courses. Microwave ovens provide a clean and cheap alternative to conventional oil baths. Most conveniently, reactions may be run at atmospheric pressure in reflux systems in ovens which have been modified to accept an appropriate condenser. As previously discussed, dipolar solvents are essential in microwave heating, and it is often necessary to adapt solvent systems in synthetic reactions to accommodate this. In substituting solvent systems for microwave synthesis, it is often the case that higher boiling point solvents are used. By doing this, it has been possible to enhance the efficiency of a number of syntheses21,22.
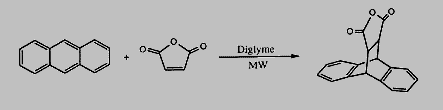
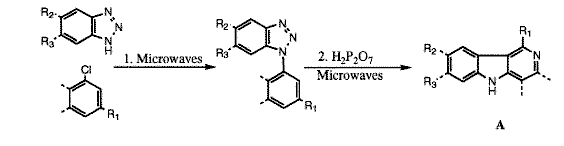
The same reaction carried out in p-xylene at between 160 and 187°c under microwave heating gave a 92% yield of the product in 3 minutes23. this compares favourably to the conventional synthesis in p-xylene at 187°c, which gave a 90% yield in 10 minutes. similar increases in reaction rate are now routinely found in homogeneous organic syntheses.24-26 Microwave techniques have offered a variety of convenient syntheses because of their cleanliness and ease of use. recent examples include amides,27 hydrogenation of [beta]-lactams,28 and 1,4 dihydropyridones.29 in the development of a series of new anti-tumour compounds, the microwave synthesis of [gamma]-carbolines (A) were possible in a one-pot reaction,30 and gave similar or higher yields than the classical method, and in a shorter period (table 1).
Table 1 Microwave acceleration of [gamma]-carboline syntheses30
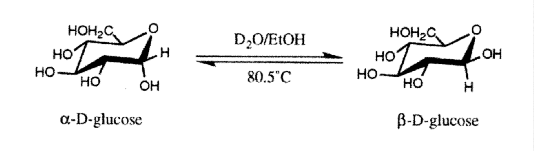
The question of whether there is a specific microwave effect which enhances organic reactions, and which cannot be attributed to superheating, is still open to debate. Examples of claims for specific activation are open to criticism in the light of what is now known about microwave induced superheating in solvents. Adamek and Hajek,31 for example, report non-thermal activation of organic reactions. By performing reactions at the same temperature under both conventional and microwave heating, they report a rate increase with microwave radiation. The rate increase is solvent dependent, suggesting that superheating may be responsible. In a related paper, Pagnotta et al.32 report a significant difference in the mutarotation of [alpha]-D-glucose to [beta]-D-glucose when performed with microwave heating rather than conventionally:
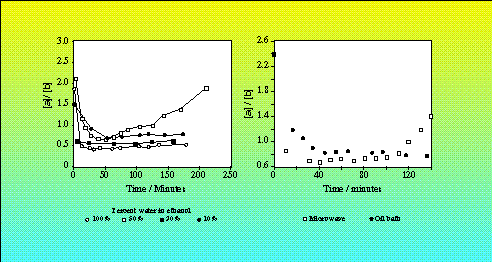
The mutarotation was carried out at 50°C and at reflux for several hours under microwave and conventional heating, and using various D2O:EtOH ratios. Superheating was suppressed in the refluxed microwave sample by the presence of a wooden applicator stick. Analysis of the a:b ratio was carried out by NMR, taking advantage of the different shifts of the anomeric hydrogens in the two forms. The results of this were rather surprising, and suggest that with a 1:1 D2O:EtOH ratio, the a:[beta] ratio increases with time under microwave heating (Figure 1a). Moreover, this effect is specific to the microwave heated sample (Figure 1b).
Figure 1. Mutarotation of a-D-Glucose Under microwave and conventional heating32
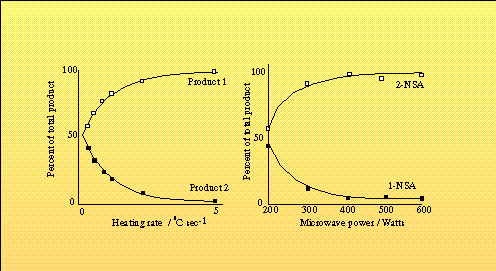
The authors suggest that the differences might be attributed to a specific microwave effect. They propose that specific activation of the polar functionalities occurs in either the glucose or the surrounding solvent cage, and that this is more effective for one form than the other. If true, this would clearly affect the equilibrium between the two forms, potentially giving rise to the plots which are observed. In a separate analysis of the effects of microwaves, Stuerga et al. suggested that a specific effect may arise purely from the increased heating rate which dielectric heating affords.33 They report that simply as a result of the more rapid heating which microwaves induce, the reaction time of their system may be cut by 75%. More important is their consideration of the kinetics of two competing reversible reactions with one common reactant. Allowing for differing activation energies and rate constants in the forward and reverse reactions, they have devised a model which predicts increasing product selectivity depending upon dT/dt (Figure 2a). Their model correlates well with the experimental results from the sulphonation of naphthalene under microwave conditions to give 1- and 2-naphthalene sulphonic acids (NSA) first considered by Abramovitch.34 The authors employed a single mode cavity to irradiate a 15cm3 sample with a range of power levels. The kinetics of this particular system result in the preferential production of 2-NSA over 1-NSA at high temperatures (~130°C), but allow production of the latter at lower temperatures, and under slow heating an almost equimolar mixture of 1- and 2-NSA is produced. Under rapid heating, the time spent by the reactants at lower temperatures is decreased, and an increasingly pure sample of the 2-NSA results (Figure 2b). The selectivity results directly from the evolution of temperature, and is independent of the heating method. Microwave heating simply enables production of the heating rates required to observe this effect. This is in line with a series of kinetic studies on esterification,35 Diels-Alder, and acid catalysed isomerisation reactions,36 which conclude that the kinetics and order of these reactions are unaffected by the method of heating (microwave or oil bath).
Figure 2 The effects of heating rate on selectivity. a) Theoretical model and b) Experimental results from the sulphonation of naphthalene
Organic Syntheses at Atmospheric Pressure The remote nature of the coupling between the microwave source and the sample allows the direct heating of solvents held in sealed microwave-transparent containers. The rate enhancements shown in reactions under microwave irradiation are increased by the effects of pressure on the solvent boiling point by allowing reactions to be conveniently run at much higher temperatures. In the earliest work in this area, Gedye37-40 and Giguere23, 41 performed a variety of reactions in screw-capped teflon vessels, the latter using the melting points of a variety of compounds to estimate the temperatures reached in the vessels. Using 150cm3 vessels for the reactions, Gedye et al. reported dramatic rate increases of up to 240 times the conventional method. Similar results were also reported by Giguere (Table 2).
Table 2 Microwave induced reactions under pressure
The volume of the vessel, solvent : space volume ratio, and solvent boiling point all affect the rate increases which are observed. Clearly, the rate increase will be greatest for those reactions normally carried out in low boiling point solvents. The flask volume, on the other hand, shows an inverse relationship to the acceleration which is observed, indicating that there is a direct relationship of the volume with the pressure which has built up inside the vessel. The solvent:space volume ratio shows a more complex relationship to the rate enhancement. The pressure build-up increases with the decrease in the space volume, but as the solvent volume increases the temperature rises more slowly, causing the rate to pass through a maximum at the optimum ratio. Knowing the optimum ratio therefore enables optimisation of the synthesis itself giving rate increases of up to 180 times the conventional synthesis. The pressure within the reaction vessel, and the associated reaction temperature increases which it allows, play an important role in the rate increases of the reactions just described and no specific microwave effects need to the invoked to describe the rate increases. In spite of this, the acid catalysed hydrolysis of diazepam to 2-methylamino-5-chlorobenzophenone,42 a reaction normally carried out at pressure in sealed glass vessels, shows a 12x rate increase under microwave conditions. Once again, there is a power dependent relationship on the rate of hydrolysis, which would clearly be in keeping with the volume:rate relationship noted by Gedye, if this is regarded as a general power per unit volume : rate relationship. At full power, the hydrolysis proceeds rapidly to completion.
The rapid expansion of microwaves in synthetic chemistry has resulted in a large range of reactions being reported under these conditions. Most of these varying only in minor details from the reactions described here. Reactions to date include hetero Diels-Alder reactions, 43 racemisation of large organic molecules via Diels-Alder cycloreversions, 44 Hydrolysis, esterification, epoxidation and cyclisation reactions.45 The processing of large quantities of material by scaling up small low volume microwave reactions is not always feasible or advantageous, nor is it always suitable for the process in question. By the use of suitable pumps and high pressure PTFE tubing, it is possible to build a flow system into the high pressure cell, and this has been put to use, for example, in the hydrolysis of formetanate to m-aminophenol.26
The reaction occurs at up to 16 times the classical thermal hydrolysis at 90°C, with the operation being suitable for continuous operation at up to 16cm3min-1. A particularly important application of the decreased reaction times which result from microwave processing is in the synthesis of radiolabelled chemicals. The location of pharmaceutical products using PET during pre-clinical trials relies on the use of a number of nuclides with very short half-lives. The commonest used isotopes for this work include122I(t1/2=3.6min.), 11 C(t1/2=20 min.), and 18F(t1/2=110 min.), and savings of even a few minutes may dramatically affect the purity of the radioisotopes in their compounds. The nucleophilic substitution of nitro- and fluoro- groups by 18F on activated benzenes has been shown to occur with much higher rates under 500Watt microwave heating.46 Samples held in 3cm3Reactivials® gave similar or improved yields to 30 minute conventional syntheses after only 5 minutes. Similarly, 131I isotope exchange reactions were completed in less than 5 minutes, with aromatic iodine compounds giving 80% completion after only one minute. The relevance of this to work with the short-lived medically useful 122I and 123I([gamma], t1/2=13.2 hrs.) isotopes is obvious. The synthesis of epi-[18F]-fluoromisonidazole, an investigative chemical for use in suspected cases of myocardial infarction, has also been improved by these methods.47 The first step, substitution of tosylate by 18F in 2cm3 Reactivials", took just 3 minutes to give a 70-80% yield. Reaction of the resulting fluorohydrin with 2-nitro-imidazole required a further 10 minutes to yield 65% of the desired product, compared to a 40% yield by the best conventional methods. The total synthesis (including working up procedures) could be accomplished in under 70 minutes to give a 40% radiochemical yield.
Pneumatic handling systems in this work reduced the radiation exposure risk in workers. The use of microwave heating flow systems similar to those already discussed may also be used for this purpose, and are found to improve the rate at which radiochemicals may be prepared. Such continuous flow systems, operating at up to 12 atm./200oC48 and with up to 20cm3min-1 of material49 are already in operation, and are capable of higher efficiency than equivalent batch systems.
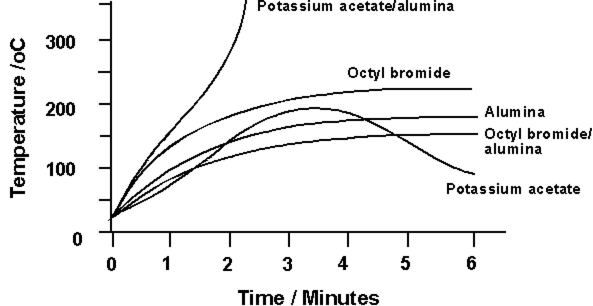
Organic Syntheses in Dry Media The largest proportion of this work in this area relates to chemical syntheses carried out on supports. By dissolving the components of a reaction in a volatile solvent, e.g. CH2Cl2, mixing the solution with a suitable solid support, and allowing the solvent to dissolve, it is then possible to activate chemical reactions on the support material by heating. Suitable support materials include Montmorillonite clays, alumina, silica, alkali metal fluoride doped alumina., and commercial Bentonitic earths The ratio of materials to this support is usually quite small - of the order of a few tens of mmol of reactant to one or two grams of support. Once the reaction has taken place, the products may be retrieved by washing the support with a suitable solvent and working up in the usual manner. Microwaves have been used extremely successfully to activate these reactions, and perhaps surprisingly, it is not always necessary for the reaction components to have high dielectric losses. In the condensation of 1-bromo-octane with potassium acetate on alumina,137 it is interesting to note that the heating characteristics of the reactants are relatively poor, whilst the alumina supported potassium acetate is capable of heating to 300°C within two minutes (Figure 3). It is heat from this process which drives the reaction.
Figure 3 Microwave heating characteristics for materials in a supported reaction (4g samples, 600Watts)137
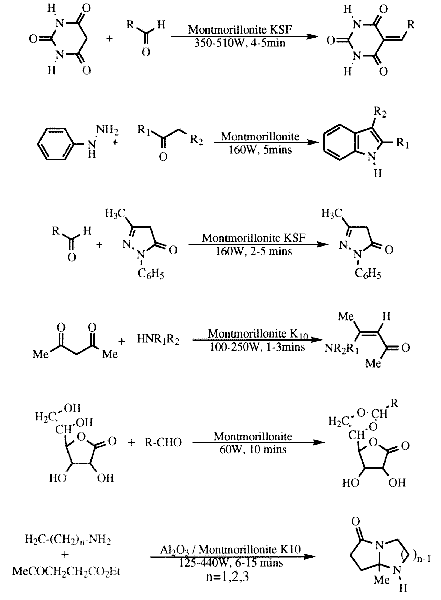
The vast majority of the support reactions which have been carried out are condensations, examples of which are listed in Table 3.
Table 3 Microwave induced reactions on supports
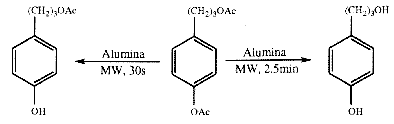
Acetylation / Deacetylation is a common method for the protection of hydroxy groups, and there are a large number of methods available for the process.144 By irradiating a protected compound held on a support, it is possible to effect rapid deacetylation within 2-3 minutes. More importantly from a synthetic perspective is the observed rate difference between aryl and alcoholic acetates;
The microwave induced reaction shows a large rate increase with respect to the conventional method. The above reaction required 40 hours at 75°C when using oil bath heating. A similar deprotection reaction may be effected when benzyl esters are used to protect carboxylic acid groups. Although a number of agents may be used for debenzylation, including catalytic or chemical hydrogenation, they provide poor yields and selectivity. By using alumina as a support for benzyl esters under microwave irradiation, rapid deacylation occurs in 10 minutes or less to give high yields of the carboxylic acids.145 As for the deprotection of OH groups, the rate of the reaction is reported to be increased for a given temperature. Because no details are given of how the temperature is measured, it is difficult to assess the validity of these claims. In recent years, dry media other than supports have been used to drive reactions under microwave irradiation, in particular p-toluene sulphonic acid (PTSA) is commonly used as a homogeneous acid. Although catalytic amounts of PTSA have been used in combination with Montmorillonite K10 to the form [beta]-aminoesters from a benzylidine aniline and a silyl ketene acetal146
When used as a co-reactant in the dry medium synthesis of esters, PTSA invariably showed yields some 9-20% higher than the best reactions on supports under identical conditions.147
REFERENCES
27. M.P. Vazquez-Tato. Synlett7, 506 (1993). 28. A.K. Bose, B.K. Banik, K.J. Barakat & M.S. Manhas. Synlett8, 575-576 (1993). 29. R. Alajarin, J.J. Vaquero, J.L.G. Navio & J. Alvarez-Builla.Synlett4, 297-298 (1992). 30. A. Molina, J.J. Vaquero, J.L. Garcia-Navio & J. Alvarez-Builla.Tetrahedron Letters34, 2673-2676 (1993). 31. F. Adamek & M. Hajek. Tetrahedron Letters33, 2039-2042 (1992). 32. M. Pagnotta, C.L.F. Pooley, B. Gurland & M. Choi. J Phys Org Chem6, 407-411 (1993). 33. D. Stuerga, K. Gonon & M. Lallemant. Tetrahedron49, 6229-6234 (1993). 34. R. Abramovitch, D. Abramovitch, K. Iyanar & K. Tamareselvy.Tetrahedron Letters32, 5251-5254 (1991). 35. K.D. Raner & C.R. Strauss. J. Org. Chem.57, 6231 (1992). 36. K.D. Raner, C.R. Strauss, F. Vyskoc & L. Mokbel. J Org Chem58, 950-953 (1993). 37. R. Gedye, et al. Tet. Lett.27, 279 (1986). 38. R. Gedye, F. Smith & K. Westaway. Educ. Chem.25, 55 (1988). 39. R.N. Gedye, F.E. Smith & K.C. Westaway. Can. J. Chem.66, 17 (1988). 40. R. Gedye, W. Rank & K. Westaway. Canadian Journal Of Chemistry-journal Canadien De Chimie69, 706-711 (1991). 41. R.J. Giguere, et al. Tet. Lett.28, 6553 (1987). 42. M.d.l. Guardia, A. Salvador, M.J. Gomez & Z.A. Debenzo. Anal. Chim. Acta224, 123 (1989). 43. A. Stambouli, M. Chastrette & M. Soufiaoui. Tetrahedron Letters32, 1723-1724 (1991). 44. S. Takano, A. Kijima, T. Sugihara, S. Satoh & K. Ogasawara.Chemistry Letters 87 (1989). 45. M.S.F. Lie Ken Jie & C. Yan kit. Lipids23, 367 (1988). 46. D.-R. Hwang, S.M. Moerlein, L. Lang & M.J. Welch. J. Che,. Soc. Chem. Commun. 1799 (1987). 47. T.J. McCarthy, C.S. Dence & M.J. Welch. Appl. Radiat. Isot.44, 1129-1132 (1993). 48. C. Peterson. New Scientist123, 44 (1989). 49. S.T. Chen, S.H. Chiou & K.T. Wang. J. Chem. Soc., Chem. Commun. 807 (1990). 137. G. Bram, A. Loupy, M. Majdoub, E. Gutierrez & E. Ruizhitzky.Tetrahedron46, 5167 (1990). 138. D. Villemin & B. Labiad. Synthetic Communications20, 3333-3337 (1990). 139. D. Villemin, B. Labiad & Y. Ouhilal. Chem. and Ind.18, 607 (1989). 140. D. Villemin & B. Labiad. Synth. Commun.20, 3213 (1990). 141. B. Rechsteiner, F. Texier-Boullet & J. Hamelin. Tet. Lett.34, 5071-5074 (1993). 142. M. Csiba, J. Cleophax, A. Loupy, J. Malthete & S.D. Gero.Tetrahedron Lett34, 1787-1790 (1993). 143. J.F. Pilard, B. Klein, F. Texierboullet & J. Hamelin. Synlett3, 219-220 (1992). 144. T.W. Greene & P.G.M. Wuts. Protective Groups in Organic Synthesis Wiley, New York (1991). 145. R.S. Varma, A.K. Chatterjee & M. Varma. Tetrahedron Lett34, 4603-4606 (1993). 146. F. Texier-Boullet, R. Latouche & J. Hamelin. Tetrahedron Lett34, 2123-2126 (1993). 147. A. Loupy, et al. Can J Chem71, 90-95 (1993). 148. G.G. Allan, B.B. Krieger & D.W. Work. J. Appl. Poly.25, 1839 (1980). 149. G. Roussy, J.-M. Thiebaut, M. Anzarmou, C. Richard & R. Martin. J. Microwave Power Electromagnetic Energy Symp. Summ. 169 (1987).
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||